Modulatory Role of Saroglitazar on Novel Hematological Inflammatory Ratios and Metabolic Parameters in Animal Model of 5-Fluorouracil Toxicity
DOI:
https://doi.org/10.54133/ajms.v10i2.2325Keywords:
Chemotherapy-induced toxicity, 5-FU, Hematological inflammatory ratios, SaroglitazarAbstract
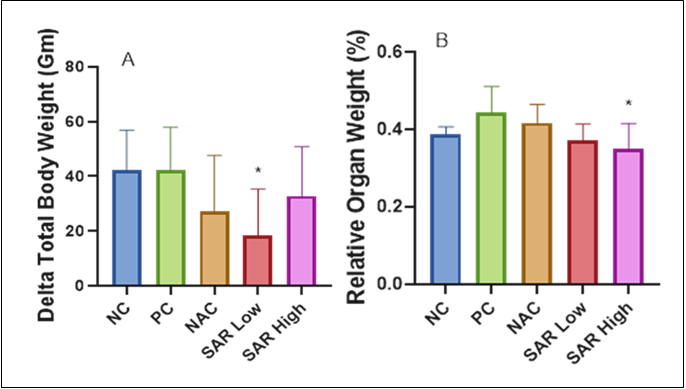
Background: 5-Fluorouracil (5-FU), a widely used chemotherapeutic agent, causes oxidative stress, inflammation, and multi-organ damage, particularly cardiotoxicity. Saroglitazar, a dual peroxisome proliferator-activated receptor α/γ agonist with lipid-lowering, insulin-sensitizing, and anti-inflammatory effects, may ameliorate these adverse outcomes. Objective: To evaluate how saroglitazar influences hematological inflammatory ratios and metabolic parameters in a rat model of 5-fluorouracil-induced toxicity. Methods: 35 adult males Wistar rats were categorized into five groups: Control (NC), 5-FU-treated positive control (PC), N-acetylcysteine (NAC) (100mg/kg) + 5-FU, SAR low (0.5mg/kg)+5-FU, and SAR high (5.0mg/kg)+5-FU. 150mg/kg 5-FU was given intraperitoneally on the 10th day of treatment. Biochemical markers were evaluated, including cholesterol, triglycerides, low-density lipoprotein (LDL), high-density lipoprotein (HDL), glucose, and HbA1c; inflammatory markers such as neutrophil-to-lymphocyte ratio (NLR), monocyte-to-lymphocyte ratio (MLR), and platelet-to-lymphocyte ratio (PLR); and hematological markers such as hemoglobin concentration (Hb), red blood cell count (RBC), and white blood cell count (WBC). Results: 5-FU treatment caused significant metabolic alteration, including dyslipidemia, hyperglycemia, systemic inflammation, and hematological suppression. Saroglitazar, on the other hand, improved lipid profiles by reducing cholesterol, triglycerides, and LDL and showed a glucose-lowering effect. It also reduced NLR and MLR, demonstrating their superior anti-inflammatory activity. Saroglitazar also improved RBC count and Hb levels, indicating its influence on erythropoiesis and bone marrow function. Conclusion: Saroglitazar mitigates 5-fluorouracil-induced metabolic, inflammatory, and hematological disturbances, supporting its potential as an adjunct to chemotherapy. Assessing its long-term effectiveness and practical use is recommended.
Downloads
References
Chrysostomou D, Roberts LA, Marchesi JR, Kinross JM. Gut microbiota modulation of efficacy and toxicity of cancer chemotherapy and immunotherapy. Gastroenterology. 2023;164(2):198-213. doi: 10.1053/j.gastro.2022.10.018.
Ali HH, Ahmed ZA, Aziz TA. Effect of telmisartan and quercetin in 5 fluorouracil-induced renal toxicity in rats. J Inflamm Res. 2022;15:6113–6124. doi: 10.2147/JIR.S389017.
da Silva MC, Fabiano LC, da Costa Salomão KC, de Freitas PLZ, Neves CQ, Borges SC, et al. A rodent model of human-dose-equivalent 5-fluorouracil: Toxicity in the liver, kidneys, and lungs. Antioxidants. 2023;12(5):1005. doi: 10.3390/antiox12051005.
Jungsuwadee P, Vore M, Clair DKS. Chemotherapy-induced oxidative stress in nontargeted normal tissues. In: Spitz DR, Dornfeld KJ, Krishnan K, Gius D, (eds.), Oxidative Stress in Cancer Biology and Therapy. Humana Press; 2012. p. 97–129. doi: 10.1007/978-1-61779-397-4_6.
Petak I, Tillman DM, Houghton JA. p53 dependence of Fas induction and acute apoptosis in response to 5-fluorouracil-leucovorin in human colon carcinoma cell lines1. Clini Cancer Res. 2000;6(11):4432–4441. PMID: 11106264.
Gelen V, Şengül E, Yıldırım S, Senturk E, Tekin S, Kükürt A. The protective effects of hesperidin and curcumin on 5-fluorouracil–induced nephrotoxicity in mice. Environ Sci Pollut Res [Internet]. 2021;28(34):47046–54705. doi: 10.1007/s11356-021-13969-5.
Kodama Y, Fumoto S, Nishi J, Nakashima M, Sasaki H, Nakamura J, et al. Absorption and distribution characteristics of 5-fluorouracil (5-FU) after an application to the liver surface in rats in order to reduce systemic side effects. Biol Pharm Bull. 2008;31(5):1049–1052. doi: 10.1248/bpb.31.1049.
Leocádio PCL, Antunes MM, Teixeira LG, Leonel AJ, Alvarez-Leite JI, Machado DCC, et al. L-arginine pretreatment reduces intestinal mucositis as induced by 5-FU in mice. Nutr Cancer. 2015;67(3):486–493. doi: 10.1080/01635581.2015.1004730.
Sara JD, Kaur J, Khodadadi R, Rehman M, Lobo R, Chakrabarti S, et al. 5-fluorouracil and cardiotoxicity: a review. Ther Adv Med Oncol. 2018;10:1758835918780140. doi: 10.1177/1758835918780140.
Madeddu C, Deidda M, Piras A, Cadeddu C, Demurtas L, Puzzoni M, et al. Pathophysiology of cardiotoxicity induced by nonanthracycline chemotherapy. J Cardiovasc Medicine. 2016;17:e12. doi: 10.2459/JCM.0000000000000376.
Jin X, Bai Y, Gao L, Wu S. Incidence of and risk factors for cardiotoxicity after fluorouracil-based chemotherapy in locally advanced or metastatic gastric cancer patients. Cancer Chemother Pharmacol. 2019;84(3):599–607. doi: 10.1007/s00280-019-03888-1.
Karakulak UN, Aladağ E, Maharjan N, Övünç K. Capecitabine-induced coronary artery vasospasm in a patient who previously experienced a similar episode with fluorouracil therapy. Turk Kardiyol Dern Ars. 2016;44(1):71-74. doi: 10.5543/tkda.2015.36005.
Yu X, Ruan Y, Shen T, Qiu Q, Yan M, Sun S, et al. Dexrazoxane Protects Cardiomyocyte from Doxorubicin-Induced Apoptosis by Modulating miR-17-5p. BioMed Res Int. 2020;2020(1):5107193. doi: 10.1155/2020/5107193.
Khan SA, Campbell AM, Lu Y, An L, Alpert JS, Chen QM. N-Acetylcysteine for cardiac protection during coronary artery reperfusion: A systematic review and meta-analysis of randomized controlled trials. Front Cardiovasc Med. 2021;8. doi: 10.3389/fcvm.2021.752939.
Pecoraro M, Marzocco S, Belvedere R, Petrella A, Franceschelli S, Popolo A. Simvastatin reduces doxorubicin-induced cardiotoxicity: Effects beyond its antioxidant activity. Int J Mol Sci. 2023;24(8):7573. doi: 10.3390/ijms24087573.
Kalliora C, Drosatos K. The Glitazars paradox: Cardiotoxicity of the metabolically beneficial dual PPARα and PPARγ activation. J Cardiovasc Pharmacol. 2020;76(5):514. doi: 10.1097/FJC.0000000000000891.
Jain N, Bhansali S, Kurpad AV, Hawkins M, Sharma A, Kaur S, et al. Effect of a dual PPAR α/γ agonist on insulin sensitivity in patients of type 2 diabetes with hypertriglyceridemia- randomized double-blind placebo-controlled trial. Sci Rep. 2019;9(1):19017. doi: 10.1038/s41598-019-55466-3.
Percie du Sert N, Hurst V, Ahluwalia A, Alam S, Avey MT, Baker M, et al. The ARRIVE guidelines 2.0: Updated guidelines for reporting animal research. J Cereb Blood Flow Metab. 2020;40(9):1769–1777. doi: 10.1177/0271678X20943823
Sharma A, S A, Kushwah DS, S R. Saroglitazar, a novel cardiometabolic agent for diabetic dyslipidemia - A Review. J Young Pharm. 2014;7(1):2–6. Available from: http://www.jyoungpharm.org/article/735
Haleagrahara N, Julian V, Chakravarthi S. N-acetylcysteine offers cardioprotection by decreasing cardiac lipid hydroperoxides and 8-isoprostane level in isoproterenol-induced cardiotoxicity in rats. Cardiovasc Toxicol. 2011;11(4):373–381. doi: 10.1007/s12012-011-9132-0.
Pan Y, Li Y, Fan H, Cui H, Chen Z, Wang Y, et al. Roles of the peroxisome proliferator-activated receptors (PPARs) in the pathogenesis of hepatocellular carcinoma (HCC). Biomed Pharmacother. 2024;177:117089. doi: 10.1016/j.biopha.2024.117089.
Botta M, Audano M, Sahebkar A, Sirtori CR, Mitro N, Ruscica M. PPAR agonists and metabolic syndrome: An established role? Int J Mol Sci. 2018;19(4):1197. doi: 10.3390/ijms19041197.
Cheng HS, Tan WR, Low ZS, Marvalim C, Lee JYH, Tan NS. Exploration and development of PPAR modulators in health and disease: An update of clinical evidence. Int J Mol Sci. 2019;20(20):5055. doi: 10.3390/ijms20205055.
Quintão NLM, Santin JR, Stoeberl LC, Corrêa TP, Melato J, Costa R. Pharmacological treatment of chemotherapy-induced neuropathic pain: PPARγ agonists as a promising tool. Front Neurosci. 2019;13. doi: 10.3389/fnins.2019.00907.
Alkabbani MA, Shatat AAS, Ghazy AN, Magdy K. Chemotherapy-induced liver injury: Unveiling emerging mechanisms and exploring mitigation strategies. ERU Res J. 2024. doi: 10.21608/erurj.2024.256957.1100.
Sun Z, Zhang S, Liang J, Li C, Yang X, Liu QS, et al. Effects of multiple novel bisphenol S analogs on adipogenesis in 3T3-L1 cells. J Hazard Mater. 2025;489:137689. doi: 10.1016/j.jhazmat.2025.137689.
Montaigne D, Butruille L, Staels B. PPAR control of metabolism and cardiovascular functions. Nat Rev Cardiol. 2021;18(12):809-823. doi: 10.1038/s41569-021-00569-6.
Sommer J, Mahli A, Freese K, Schiergens TS, Kuecuekoktay FS, Teufel A, et al. Analysis of molecular mechanisms of 5-fluorouracil-induced steatosis and inflammation in vitro and in mice. Oncotarget. 2017;8(8):13059-13072. doi: 10.18632/oncotarget.14371.
Trehan S, Singh G, Singh A, Bector G, Jain A, Antil P, et al. Chemotherapy and metabolic syndrome: A comprehensive review of molecular pathways and clinical outcomes. Cureus. 2024;16(8):e66354. doi: 10.7759/cureus.66354.
Jain MR, Giri SR, Trivedi C, Bhoi B, Rath A, Vanage G, et al. Saroglitazar, a novel PPARα/γ agonist with predominant PPARα activity, shows lipid-lowering and insulin-sensitizing effects in preclinical models. Pharmacol Res Perspect. 2015;3(3):e00136. doi: 10.1002/prp2.136.
Niu Y, Xiao H, Wang B, Wang Z, Du K, Wang Y, et al. Angelica sinensis polysaccharides alleviate the oxidative burden on hematopoietic cells by restoring 5-fluorouracil-induced oxidative damage in perivascular mesenchymal progenitor cells. Pharm Biol. 2023;61(1):768–778. doi: 10.1080/13880209.2023.2207592.
Fougerat A, Montagner A, Loiseau N, Guillou H, Wahli W. Peroxisome proliferator-activated receptors and their novel ligands as candidates for the treatment of non-alcoholic fatty liver disease. Cells. 2020;9(7):1638. doi: 10.3390/cells9071638.
Joharapurkar AA, Pandya VB, Patel VJ, Desai RC, Jain MR. Prolyl hydroxylase inhibitors: A breakthrough in the therapy of anemia associated with chronic diseases. J Med Chem. 2018;61(16):6964–6982. doi: 10.1021/acs.jmedchem.7b01686.
Wang Z, Wang Q, Gong X. Unveiling the mysteries of contrast-induced acute kidney injury: New horizons in pathogenesis and prevention. Toxics. 2024;12(8):620. doi: 10.3390/toxics12080620.
Balakumar P, Mahadevan N, Sambathkumar R. A contemporary overview of PPARα/γ dual agonists for the management of diabetic dyslipidemia. Curr Mol Pharmacol. 2019;12(3):195-201. doi: 10.2174/1874467212666190111165015.
Downloads
Published
How to Cite
Issue
Section
License
Copyright (c) 2026 Al-Rafidain Journal of Medical Sciences ( ISSN 2789-3219 )

This work is licensed under a Creative Commons Attribution-NonCommercial-ShareAlike 4.0 International License.
Published by Al-Rafidain University College. This is an open access journal issued under the CC BY-NC-SA 4.0 license (https://creativecommons.org/licenses/by-nc-sa/4.0/).











